
Base Year Value ()
CAGR ()
Forecast Year Value ()
Historical Data Period
Largest Region
Forecast Period
Mercado de diagnósticos complementarios por productos (ensayos, kits y reactivos, software y servicios), por tecnología (reacción en cadena de la polimerasa, secuenciación de próxima generación, hibridación in situ, inmunohistoquímica), por indicación (cáncer, enfermedades neurológicas, enfermedades infecciosas, enfermedades cardiovasculares), por usuario final (empresas farmacéuticas y biofarmacéuticas, organizaciones de investigación por contrato, laboratorios) y región, tendencias globales y pronóstico de 2022 a 2029
Instant access to hundreds of data points and trends
- Market estimates from 2014-2029
- Competitive analysis, industry segmentation, financial benchmarks
- Incorporates SWOT, Porter's Five Forces and risk management frameworks
- PDF report or online database with Word, Excel and PowerPoint export options
- 100% money back guarantee
Descripción general del mercado de diagnóstico complementario
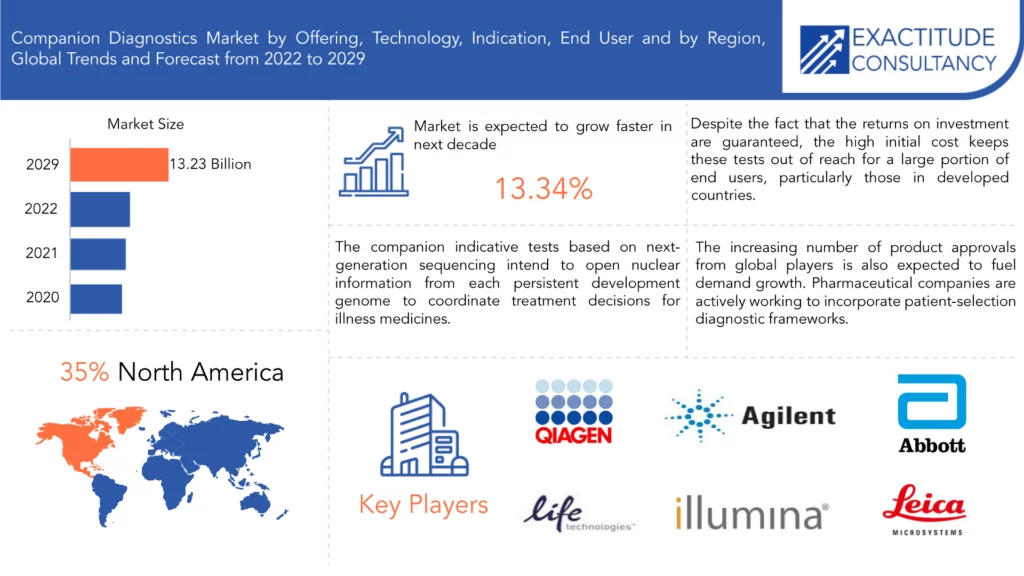
Se espera que el mercado de diagnósticos complementarios crezca a una tasa anual compuesta del 13,34 % entre 2021 y 2029. Se espera que alcance más de 13 230 millones de dólares en 2029 desde los 3500 millones de dólares en 2020.
Los diagnósticos complementarios son pruebas in vitro que brindan información sobre las respuestas terapéuticas de los pacientes a un tratamiento específico. Se trata de un dispositivo médico in vitro que brinda información necesaria para el manejo seguro y eficaz de un medicamento o producto biológico correspondiente. Esta prueba diagnóstica ayuda al médico a determinar si un producto terapéutico específico es beneficioso para los pacientes y puede compensar cualquier posible efecto secundario o riesgo grave.
El aumento de los distintos tipos de cáncer entre las personas es el principal factor que impulsa el crecimiento del mercado. A medida que aumenta el número de ensayos clínicos y la demanda de secuenciación de última generación , también lo hará el mercado de diagnóstico complementario. Otro factor que impulsa dicho crecimiento es la importancia de los productos de este mercado en la producción de diversos tipos de medicamentos.
Se espera que el mercado de diagnósticos complementarios crezca significativamente durante el período de pronóstico, debido a un aumento en la I+D de terapias dirigidas, un aumento en la demanda de medicina de precisión con creciente atención en las economías emergentes, el descubrimiento de nuevos biomarcadores para diversas afecciones y un mayor número de necesidades insatisfechas para el tratamiento del cáncer.
Con los avances en la secuenciación hereditaria y la genómica, ahora se acepta ampliamente que los medicamentos pueden generar resultados variables en diferentes personas. Una mejor comprensión de las características hereditarias o los biomarcadores de un individuo puede hacer avanzar el acto de supervisar "la medicación correcta, en el momento perfecto, en la dosis correcta, para el individuo perfecto". Las compañías farmacéuticas y biofarmacéuticas trabajan constantemente para implementar sistemas analíticos de elección del paciente en las primeras etapas del desarrollo de medicamentos con el fin de proporcionar tratamientos específicos al competidor adecuado. Esto contribuye al crecimiento del mercado de diagnósticos complementarios.
| ATRIBUTO | DETALLES |
| Periodo de estudio | 2020-2029 |
| Año base | 2022 |
| Año estimado | 2022 |
| Año pronosticado | 2022-2029 |
| Periodo histórico | 2018-2020 |
| Unidad | Valor (miles de millones de USD) |
| Segmentación | Por oferta, por tecnología, por indicación, por usuario final, por región |
| Por tecnología de resina |
|
| Por tecnología |
|
| Por indicación |
|
| Por el usuario final |
|
| Por región |
|
La introducción de la COVID-19 ha obstaculizado el consumo de diagnósticos complementarios. Además, los principales actores del mercado sufrieron una caída significativa de los ingresos como resultado de la pandemia de COVID-19, debido a una reducción en el volumen de pruebas y otros factores. En lo que respecta a los ingresos, el mercado creció un 15,1% en 2020, en comparación con el 19,8% en 2019.
Los ingresos generados por los principales actores disminuyeron significativamente en 2020. Por ejemplo, Myriad Genetics, Inc. informó una disminución del 34,5 % en los ingresos por diagnóstico molecular en 2020 en comparación con 2019. Además, Abbott Laboratories informó una disminución del 4,0 % en los ingresos por diagnósticos de laboratorio básicos en 2020 en comparación con 2019.
Sin embargo, el levantamiento de las restricciones de confinamiento, las órdenes de quedarse en casa en muchos países, la introducción de nuevas pruebas de diagnóstico y el restablecimiento de la red de la cadena de suministro fueron factores clave en la recuperación de las pruebas en 2021. Como resultado, la reanudación de las pruebas de diagnóstico del cáncer y la introducción de nuevas pruebas de diagnóstico impulsaron la demanda en 2021.
[título id="attachment_14559" align="aligncenter" width="1024"]
Frequently Asked Questions
• What is the worth of companion diagnostics market?
The companion diagnostics market size had crossed USD 3.5 Billion in 2020 and will observe a CAGR of more than 13.34% up to 2029.
• What are some of the market's driving forces?
Major factors driving the companion diagnostics market growth include the significant growth of companion diagnostics in drug development and rising incidence of cancer and growing adoption of targeted therapies.
• Which are the top companies to hold the market share in companion diagnostics market?
Qiagen, Agilent Technologies, Roche, Abbott Laboratories, Inc., BioMerieux, Ventana Medical Systems, Myriad Genetics, Inc., Resonance Health Ltd, Leica Microsystems, Life Technologies, F. Hoffmann-L Roche Ltd., Illumina, Inc., Guardant Health, Thermo Fisher Scientific Inc., Danaher Corporation are the major companion diagnostics market players.
• Which is the largest regional market for companion diagnostics market?
The region's largest share is in North America. Products manufactured in nations like US and Canada that perform similarly and are inexpensively accessible to the general public have led to the increasing appeal.
Companion Diagnostics Market Segment Analysis
The companion diagnostics market is segmented based on products, technology, indication, end user, and region, global trends and forecast.
On the basis of products, the market is segmented into assays, kits & reagents, software & services. Due to increase in demand for kits and reagents, the assays, kits and reagents segment is estimated to lead.
On the basis of technology, the market is segmented into polymerase chain reaction, next-generation sequencing, in situ hybridization, immunohistochemistry.
Based on technology, the pcr segment currently dominates the global market and is expected to do so throughout the forecast period. Cost effectiveness, high sensitivity, and specificity, which can be used for simple automated platforms, are some key factors driving market growth. Furthermore, the pcr, which is another key driver of this segment, can determine the sequencing of unknown etiologies of many diseases. However, the next generation sequencing segment is expected to grow rapidly during the forecast period due to key advantages such as higher sensitivity to detect low-frequency variants, shorter turnaround time for large sample volumes, the ability to sequence hundreds to thousands of genes or gene regions simultaneously, and so on.
On the basis of indication, the market is segmented into cancer, neurological diseases, infectious diseases, cardiovascular diseases.
The cancer segment is currently the largest revenue generator and is expected to grow significantly during the forecast period. Some key factors driving the growth of the cancer segment include increased cancer prevalence, increased r&d activities for cancer, increased unmet needs for cancer treatment, increased number of fda approved companion diagnostics, and increased patient awareness of personalized medicine.
Cancer research has discovered significant differences in gene sequence and expression patterns that can serve as the foundation for targeted therapy. Furthermore, many biomarkers for various cancers have been identified, for which companion diagnostics have been developed. Companion diagnostics are widely used in various types of cancer, including lung cancer, colorectal cancer, breast cancer, and blood cancer, which are the leading causes of death.
On the basis of end user, the market is segmented into pharmaceutical & biopharmaceutical companies, contract research organizations, laboratories.
There is no denying that contract research organizations (cros) are becoming more important in the development of companion diagnostics. As the demand for personalized medicine grows, cros are uniquely positioned to assist pharmaceutical and biotechnology companies in developing customized treatments based on a patient's unique genetic makeup.
Companion diagnostics are diagnostic tests used to identify patients who are likely to benefit from a specific treatment. They can also be used to track a patient's response to therapy and make necessary adjustments. Cancer, cardiovascular disease, and infectious diseases are just a few of the conditions that can be treated with companion diagnostics.
[caption id="attachment_14563" align="aligncenter" width="1920"]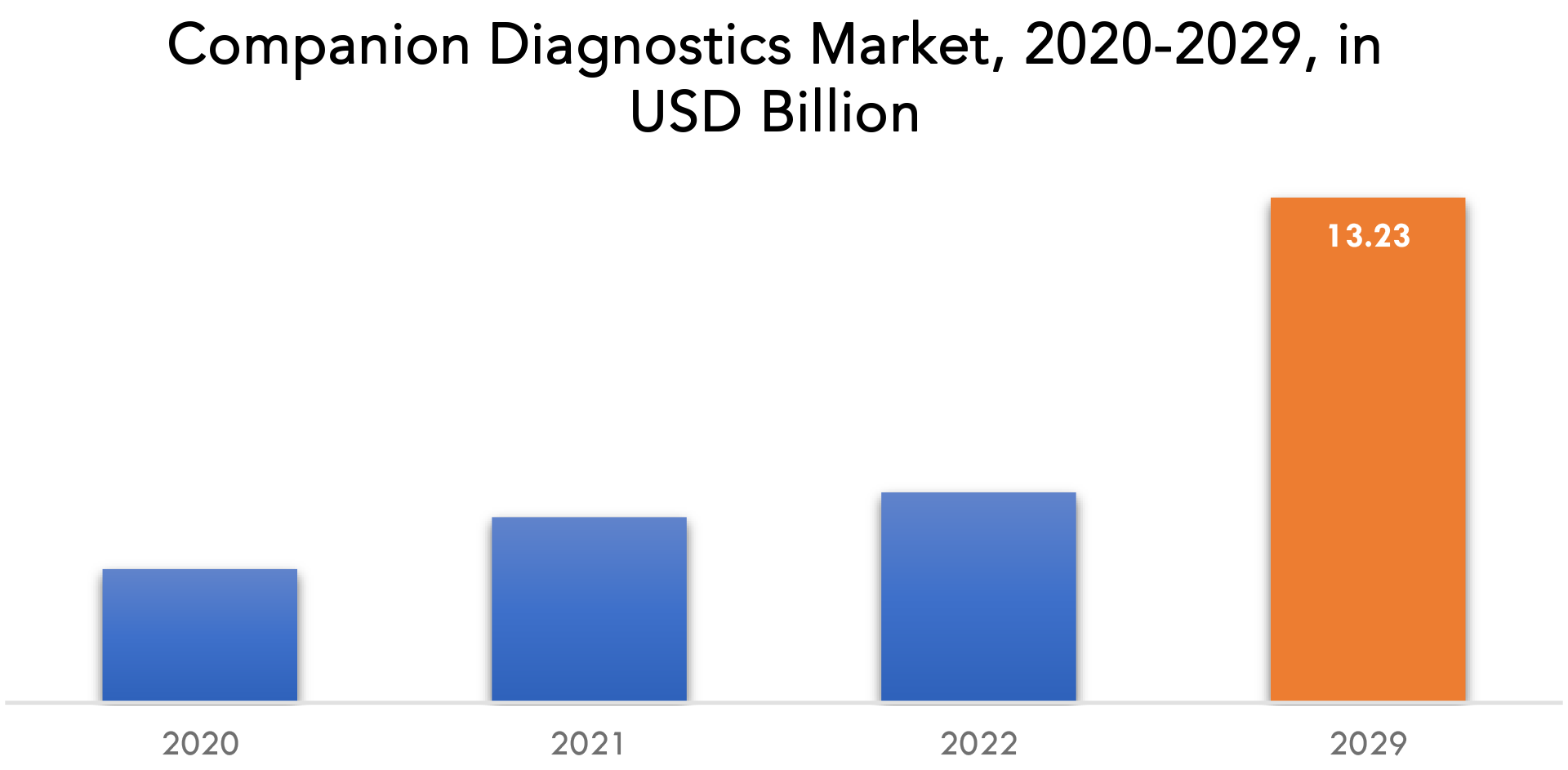
Companion Diagnostics Market Players
Qiagen, Agilent Technologies, Roche, Abbott Laboratories, Inc., BioMerieux, Ventana Medical Systems, Myriad Genetics, Inc., Resonance Health Ltd, Leica Microsystems, Life Technologies, F. Hoffmann-L Roche Ltd., Illumina, Inc., Guardant Health, Thermo Fisher Scientific Inc., Danaher Corporation are the major Companion Diagnostics market players. Recent Developments: 23 January, 2023: QIAGEN Digital Insights announced launch of their enhanced QIAGEN CLC Genomics Workbench Premium that removes the data-analysis bottleneck of Next-Generation Sequencing by adding analysis speed to analyze and interpret Whole Genome Sequencing, Whole Exome Sequencing and Large Panel Sequencing data. 9 January, 2023: QIAGEN announced of having been completed the acquisition of Verogen, a leader in the use of Next-Generation Sequencing technologies to drive the future of human identification and forensic investigation.Who Should Buy? Or Key stakeholders
- Pharmaceutical Companies
- Research and Development (R&D) Companies
- Diagnostic Laboratories
- Government Research Institute
- Academic Institutes and Universities
- Investors
- Regulatory Authorities
Companion Diagnostics Market Regional Analysis
The Companion Diagnostics Market by region includes North America, Asia-Pacific (APAC), Europe, South America, and Middle East & Africa (MEA).
- North America: includes the US, Canada, Mexico
- Asia Pacific: includes China, Japan, South Korea, India, Australia, ASEAN and Rest of APAC
- Europe: includes UK, Germany, France, Italy, Spain, Russia, and Rest of Europe
- South America: includes Brazil, Argentina and Rest of South America
- Middle East & Africa: includes Turkey, UAE, Saudi Arabia, South Africa, and Rest of MEA
North America is expected to lead the global companion diagnostics market. This could explain the high incidence rate of cancer and other chronic diseases, as well as the rising cost of healthcare in the area. Furthermore, market development is expected to drive an increase in the number of healthcare organisations working on generated genomic databases to better understand the human genome, as well as increased research activities in this area using companion diagnostic kits.
The European market for companion diagnostics is expected to be the second-largest during the review period. The expansion of the regional industry is expected to fuel the participation of a large number of companies as well as increased research and development activities aimed at improving personalised drug treatment for cancer patients. Furthermore, rising per capita income increases the preference for personalised treatment, fueling further consumer growth.
Because of the rapidly increasing number of cancer, cardiovascular, and neurological patients, Asia-Pacific is expected to be the fastest growing region. Furthermore, the rising prevalence of infectious diseases has a positive impact on business development. The high prevalence of cancer, increasing proteomics and genomics research, rising research funding, increasing investments by pharmaceutical and biotechnology firms, and rising awareness of personalised therapeutics in many APAC countries are expected to drive the growth of the APAC companion diagnostics market.
The Middle East and Africa market is expected to grow steadily due to an increase in cancer cases and a growing preference for customised drugs. The Middle East and Africa market is expected to grow steadily due to an increase in the number of hospitals and research labs, increased investment by healthcare firms, an increase in cancer cases, and increased drug discovery.
[caption id="attachment_14564" align="aligncenter" width="1920"]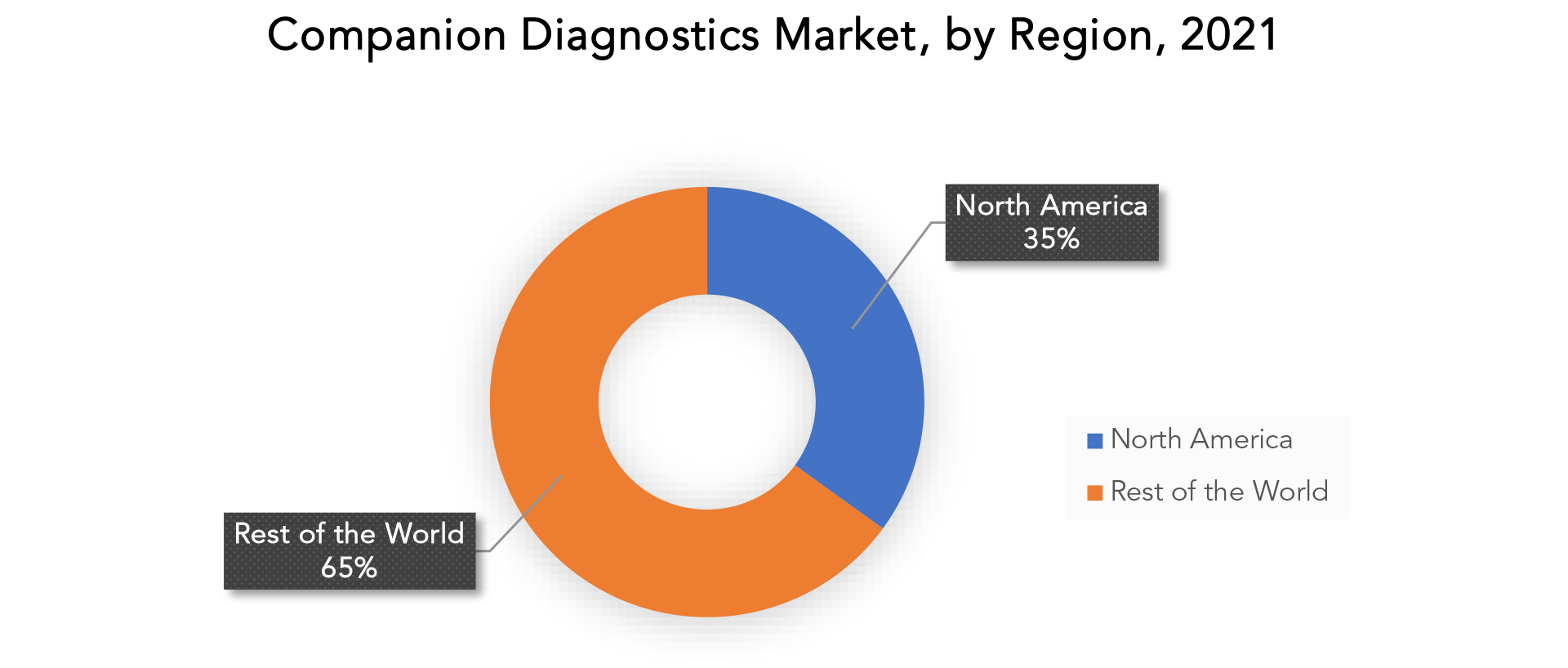
Key Market Segments: Companion Diagnostics Market
- Companion Diagnostics Market By Offering 2020-2029, (Usd Billion)
- Assays, Kits & Reagents
- Software & Services
- Polymerase Chain Reaction
- Next-Generation Sequencing
- In Situ Hybridization
- Immunohistochemistry
- Cancer
- Neurological Diseases
- Infectious Diseases
- Cardiovascular Diseases
- Pharmaceutical & Biopharmaceutical Companies
- Contract Research Organizations
- Laboratories
- North America
- Asia Pacific
- Europe
- South America
- Middle East And Africa
Exactitude Consultancy Core Companion Diagnostics Services Key Objectives:
- Increasing sales and market share
- Developing new technology
- Improving profitability
- Entering new markets
- Enhancing brand reputation
Key Question Answered
- What is the expected growth rate of the companion diagnostics market over the next 7 years?
- Who are the major players in the companion diagnostics market and what is their market share?
- What are the end-user industries driving demand for market and what is their outlook?
- What are the opportunities for growth in emerging markets such as Asia-Pacific, Middle East, and Africa?
- How is the economic environment affecting the companion diagnostics market, including factors such as interest rates, inflation, and exchange rates?
- What is the expected impact of government policies and regulations on the companion diagnostics market?
- What is the current and forecasted size and growth rate of the global companion diagnostics market?
- What are the key drivers of growth in the companion diagnostics market?
- What are the distribution channels and supply chain dynamics in the companion diagnostics market?
- What are the technological advancements and innovations in the companion diagnostics market and their impact on product development and growth?
- What are the regulatory considerations and their impact on the market?
- What are the challenges faced by players in the companion diagnostics market and how are they addressing these challenges?
- What are the opportunities for growth and expansion in the companion diagnostics market?
- What are the service offerings and specifications of leading players in the market?
- INTRODUCCIÓN
- DEFINICIÓN DE MERCADO
- SEGMENTACIÓN DEL MERCADO
- CRONOGRAMAS DE INVESTIGACIÓN
- SUPUESTOS Y LIMITACIONES
- METODOLOGÍA DE LA INVESTIGACIÓN
- MINERÍA DE DATOS
- INVESTIGACIÓN SECUNDARIA
- INVESTIGACIÓN PRIMARIA
- ASESORAMIENTO DE EXPERTOS EN LA MATERIA
- CONTROLES DE CALIDAD
- REVISIÓN FINAL
- TRIANGULACIÓN DE DATOS
- ENFOQUE DE ABAJO HACIA ARRIBA
- ENFOQUE DE ARRIBA HACIA ABAJO
- FLUJO DE INVESTIGACIÓN
- FUENTES DE DATOS
- MINERÍA DE DATOS
- RESUMEN EJECUTIVO
- PANORAMA DEL MERCADO
- PERSPECTIVA DEL MERCADO GLOBAL DE DIAGNÓSTICOS COMPLEMENTARIOS
- IMPULSORES DEL MERCADO
- RESTRICCIONES DEL MERCADO
- OPORTUNIDADES DE MERCADO
- IMPACTO DEL COVID-19 EN EL MERCADO DE DIAGNÓSTICOS COMPAÑERISMOS
- MODELO DE LAS CINCO FUERZAS DE PORTER
- AMENAZA DE NUEVOS INGRESANTES
- AMENAZA DE SUSTITUTOS
- PODER DE NEGOCIACIÓN DE LOS PROVEEDORES
- PODER DE NEGOCIACIÓN DE LOS CLIENTES
- GRADO DE COMPETENCIA
- ANÁLISIS DE LA CADENA DE VALOR DE LA INDUSTRIA
- PERSPECTIVA DEL MERCADO GLOBAL DE DIAGNÓSTICOS COMPLEMENTARIOS
- MERCADO GLOBAL DE DIAGNÓSTICOS COMPAÑEROS POR OFERTA (MIL MILLONES DE USD), 2020-2029
- ENSAYOS, KITS Y REACTIVOS
- SOFTWARE Y SERVICIOS
- SERVICIOS BÁSICOS DE DIAGNÓSTICO COMPLEMENTARIO
- MERCADO GLOBAL DE DIAGNÓSTICOS COMPLEMENTARIOS POR TECNOLOGÍA (MIL MILLONES DE USD), 2020-2029
- REACCIÓN EN CADENA DE LA POLIMERASA
- SECUENCIACIÓN DE ÚLTIMA GENERACIÓN
- HIBRIDACIÓN IN SITU
- INMUNOHISTOQUÍMICA
- MERCADO GLOBAL DE DIAGNÓSTICOS COMPLEMENTARIOS POR INDICACIÓN (MIL MILLONES DE USD), 2020-2029
- CÁNCER
- ENFERMEDADES NEUROLÓGICAS
- ENFERMEDADES INFECCIOSAS
- ENFERMEDADES CARDIOVASCULARES
- GLOBAL COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION), 2020-2029
- PHARMACEUTICAL & BIOPHARMACEUTICAL COMPANIES
- CONTRACT RESEARCH ORGANIZATIONS
- LABORATORIES
- GLOBAL COMPANION DIAGNOSTICS MARKET BY REGION (USD BILLION), 2020-2029
- NORTH AMERICA
- US
- CANADA
- MEXICO
- SOUTH AMERICA
- BRAZIL
- ARGENTINA
- COLOMBIA
- REST OF SOUTH AMERICA
- EUROPE
- GERMANY
- UK
- FRANCE
- ITALY
- SPAIN
- RUSSIA
- REST OF EUROPE
- ASIA PACIFIC
- INDIA
- CHINA
- JAPAN
- SOUTH KOREA
- AUSTRALIA
- SOUTH-EAST ASIA
- REST OF ASIA PACIFIC
- MIDDLE EAST AND AFRICA
- UAE
- SAUDI ARABIA
- SOUTH AFRICA
- REST OF MIDDLE EAST AND AFRICA
- NORTH AMERICA
- COMPANY PROFILES* (BUSINESS OVERVIEW, COMPANY SNAPSHOT, PRODUCT OFFERED, RECENT DEVELOPMENTS)
- QIAGEN
- AGILENT TECHNOLOGIES
- ROCHE
- ABBOTT LABORATORIES, INC.
- BIOMERIEUX
- VENTANA MEDICAL SYSTEMS
- MYRIAD GENETICS, INC.
- RESONANCE HEALTH LTD
- LEICA MICROSYSTEMS
- LIFE TECHNOLOGIES
- HOFFMANN-LA ROCHE LTD.
- ILLUMINA, INC.
- GUARDANT HEALTH
- THERMO FISHER SCIENTIFIC INC.
- DANAHER CORPORATION *THE COMPANY LIST IS INDICATIVE
LIST OF TABLES
TABLE 1 GLOBAL COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 2 GLOBAL COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 3 GLOBAL COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 4 GLOBAL COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 5 GLOBAL COMPANION DIAGNOSTICS MARKET BY REGION (USD BILLION) 2020-2029
TABLE 6 NORTH AMERICA COMPANION DIAGNOSTICS MARKET BY COUNTRY (USD BILLION) 2020-2029
TABLE 7 NORTH AMERICA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 8 NORTH AMERICA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 9 NORTH AMERICA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 10 NORTH AMERICA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 11 US COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 12 US COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 13 US COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 14 US COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 15 CANADA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 16 CANADA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 17 CANADA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 18 CANADA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 19 MEXICO COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 20 MEXICO COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 21 MEXICO COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 22 MEXICO COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 23 SOUTH AMERICA COMPANION DIAGNOSTICS MARKET BY COUNTRY (USD BILLION) 2020-2029
TABLE 24 SOUTH AMERICA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 25 SOUTH AMERICA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 26 SOUTH AMERICA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 27 SOUTH AMERICA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 28 BRAZIL COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 29 BRAZIL COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 30 BRAZIL COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 31 BRAZIL COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 32 ARGENTINA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 33 ARGENTINA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 34 ARGENTINA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 35 ARGENTINA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 36 COLOMBIA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 37 COLOMBIA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 38 COLOMBIA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 39 COLOMBIA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 40 REST OF SOUTH AMERICA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 41 REST OF SOUTH AMERICA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 42 REST OF SOUTH AMERICA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 43 REST OF SOUTH AMERICA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 44 ASIA-PACIFIC COMPANION DIAGNOSTICS MARKET BY COUNTRY (USD BILLION) 2020-2029
TABLE 45 ASIA-PACIFIC COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 46 ASIA-PACIFIC COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 47 ASIA-PACIFIC COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 48 ASIA-PACIFIC COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 49 INDIA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 50 INDIA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 51 INDIA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 52 INDIA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 53 CHINA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 54 CHINA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 55 CHINA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 56 CHINA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 57 JAPAN COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 58 JAPAN COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 59 JAPAN COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 60 JAPAN COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 61 SOUTH KOREA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 62 SOUTH KOREA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 63 SOUTH KOREA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 64 SOUTH KOREA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 65 AUSTRALIA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 66 AUSTRALIA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 67 AUSTRALIA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 68 AUSTRALIA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 69 SOUTH-EAST ASIA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 70 SOUTH-EAST ASIA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 71 SOUTH-EAST ASIA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 72 SOUTH-EAST ASIA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 73 REST OF ASIA PACIFIC COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 74 REST OF ASIA PACIFIC COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 75 REST OF ASIA PACIFIC COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 76 REST OF ASIA PACIFIC COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 77 EUROPE COMPANION DIAGNOSTICS MARKET BY COUNTRY (USD BILLION) 2020-2029
TABLE 78 ASIA-PACIFIC COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 79 ASIA-PACIFIC COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 80 ASIA-PACIFIC COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 81 ASIA-PACIFIC COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 82 GERMANY COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 83 GERMANY COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 84 GERMANY COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 85 GERMANY COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 86 UK COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 87 UK COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 88 UK COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 89 UK COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 90 FRANCE COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 91 FRANCE COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 92 FRANCE COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 93 FRANCE COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 94 ITALY COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 95 ITALY COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 96 ITALY COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 97 ITALY COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 98 SPAIN COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 99 SPAIN COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 100 SPAIN COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 101 SPAIN COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 102 RUSSIA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 103 RUSSIA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 104 RUSSIA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 105 RUSSIA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 106 REST OF EUROPE COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 107 REST OF EUROPE COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 108 REST OF EUROPE COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 109 REST OF EUROPE COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 110 MIDDLE EAST AND AFRICA COMPANION DIAGNOSTICS MARKET BY COUNTRY (USD BILLION) 2020-2029
TABLE 111 MIDDLE EAST AND AFRICA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 112 MIDDLE EAST AND AFRICA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 113 MIDDLE EAST AND AFRICA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 114 MIDDLE EAST AND AFRICA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 115 UAE COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 116 UAE COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 117 UAE COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 118 UAE COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 119 SAUDI ARABIA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 120 SAUDI ARABIA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 121 SAUDI ARABIA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 122 SAUDI ARABIA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 123 SOUTH AFRICA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 124 SOUTH AFRICA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 125 SOUTH AFRICA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 126 SOUTH AFRICA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
TABLE 127 REST OF MIDDLE EAST AND AFRICA COMPANION DIAGNOSTICS MARKET BY OFFERING (USD BILLION) 2020-2029
TABLE 128 REST OF MIDDLE EAST AND AFRICA COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY (USD BILLION) 2020-2029
TABLE 129 REST OF MIDDLE EAST AND AFRICA COMPANION DIAGNOSTICS MARKET BY INDICATION (USD BILLION) 2020-2029
TABLE 130 REST OF MIDDLE EAST AND AFRICA COMPANION DIAGNOSTICS MARKET BY END USER (USD BILLION) 2020-2029
LIST OF FIGURES
FIGURE 1 MARKET DYNAMICS
FIGURE 2 REPORT TIMELINES: YEARS CONSIDERED
FIGURE 3 BOTTOM-UP APPROACH
FIGURE 4 RESEARCH FLOW
FIGURE 5 GLOBAL COMPANION DIAGNOSTICS MARKET BY OFFERING USD BILLION, 2020-2029
FIGURE 6 GLOBAL COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY, USD BILLION, 2020-2029
FIGURE 7 GLOBAL COMPANION DIAGNOSTICS MARKET BY APPLICATION, USD BILLION, 2020-2029
FIGURE 8 GLOBAL COMPANION DIAGNOSTICS MARKET BY END USER, USD BILLION, 2020-2029
FIGURE 9 GLOBAL COMPANION DIAGNOSTICS MARKET BY REGION, USD BILLION, 2020-2029
FIGURE 10 PORTER’S FIVE FORCES MODEL
FIGURE 11 GLOBAL COMPANION DIAGNOSTICS MARKET BY OFFERING, USD BILLION, 2021
FIGURE 12 GLOBAL COMPANION DIAGNOSTICS MARKET BY TECHNOLOGY, USD BILLION, 2021
FIGURE 13 GLOBAL COMPANION DIAGNOSTICS MARKET BY APPLICATION, USD BILLION, 2021
FIGURE 14 GLOBAL COMPANION DIAGNOSTICS MARKET BY END USER, USD BILLION, 2021
FIGURE 15 GLOBAL COMPANION DIAGNOSTICS MARKET BY REGION, USD BILLION, 2021
FIGURE 16 MARKET SHARE ANALYSIS
FIGURE 17 QIAGEN: COMPANY SNAPSHOT
FIGURE 18 AGILENT TECHNOLOGIES: COMPANY SNAPSHOT
FIGURE 19 ROCHE: COMPANY SNAPSHOT
FIGURE 20 ABBOTT LABORATORIES, INC.: COMPANY SNAPSHOT
FIGURE 21 BIOMERIEUX: COMPANY SNAPSHOT
FIGURE 22 VENTANA MEDICAL SYSTEMS: COMPANY SNAPSHOT
FIGURE 23 MYRIAD GENETICS, INC.: COMPANY SNAPSHOT
FIGURE 24 RESONANCE HEALTH LTD: COMPANY SNAPSHOT
FIGURE 25 LEICA MICROSYSTEMS: COMPANY SNAPSHOT
FIGURE 26 LIFE TECHNOLOGIES: COMPANY SNAPSHOT
FIGURE 27 F. HOFFMANN-L ROCHE LTD.: COMPANY SNAPSHOT
FIGURE 28 ILLUMINA, INC.: COMPANY SNAPSHOT
FIGURE 29 GUARDANT HEALTH: COMPANY SNAPSHOT
FIGURE 30 THERMO FISHER SCIENTIFIC INC.: COMPANY SNAPSHOT
FIGURE 31 DANAHER CORPORATION: COMPANY SNAPSHOT
DOWNLOAD FREE SAMPLE REPORT
License Type
SPEAK WITH OUR ANALYST
Want to know more about the report or any specific requirement?
WANT TO CUSTOMIZE THE REPORT?
Our Clients Speak
We asked them to research ‘ Equipment market’ all over the world, and their whole arrangement was helpful to us. thehealthanalytics.com insightful analysis and reports contributed to our current analysis and in creating a future strategy. Besides, the te
Yosuke Mitsui
Senior Associate Construction Equipment Sales & Marketing
We asked them to research ‘Equipment market’ all over the world, and their whole arrangement was helpful to us. thehealthanalytics.com insightful analysis and reports contributed to our current analysis and in creating a future strategy. Besides, the te






