
Base Year Value ()
CAGR ()
Forecast Year Value ()
Historical Data Period
Largest Region
Forecast Period
Mercato dei controlli di qualità molecolari per prodotto (indipendente, specifico dello strumento (PCR, sequenziamento del DNA), applicazione (diagnosi delle malattie infettive, test oncologici, test genetici, altri), tipo di analita (controlli di analita singolo, controlli di analita multiplo), utente finale (laboratori diagnostici, ospedali, produttori di IVD e CRO, istituti accademici e di ricerca, altri) e regioni: previsioni globali dal 2022 al 2029
Instant access to hundreds of data points and trends
- Market estimates from 2014-2029
- Competitive analysis, industry segmentation, financial benchmarks
- Incorporates SWOT, Porter's Five Forces and risk management frameworks
- PDF report or online database with Word, Excel and PowerPoint export options
- 100% money back guarantee
Panoramica del mercato dei controlli di qualità molecolari
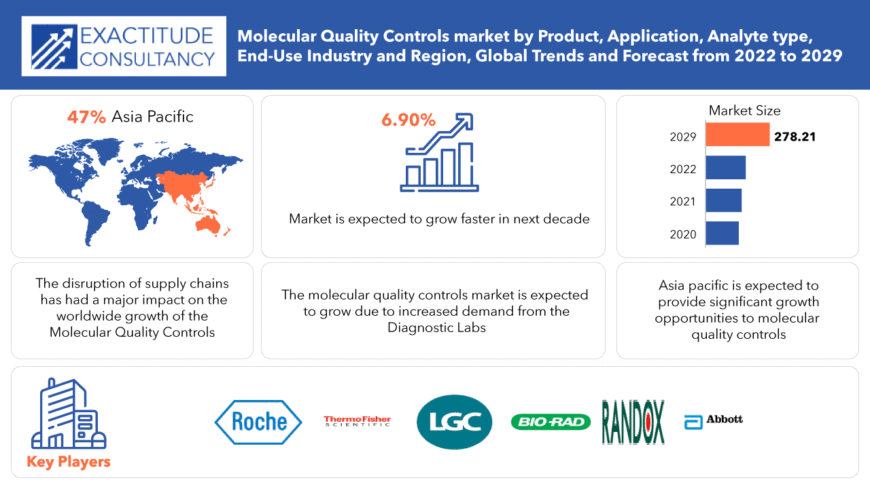
Il mercato globale dei controlli di qualità molecolari ammonta a 154,43 miliardi di USD nel 2020 e si prevede che crescerà fino a 278,21 miliardi di USD entro il 2029, con un tasso di crescita annuale composto (CAGR) del 6,90%.
Il mercato dei controlli di qualità molecolari comprende uno spettro di prodotti progettati per sostenere la precisione e l'affidabilità dei test diagnostici molecolari attraverso la fornitura di materiali standardizzati per la garanzia della qualità e i test di competenza. Questi controlli sono impiegati per supervisionare l'efficacia dei test molecolari, comprendendo tecniche come la reazione a catena della polimerasi (PCR), il sequenziamento di nuova generazione (NGS) e altre metodologie basate sugli acidi nucleici , all'interno di diversi contesti di laboratorio , tra cui diagnostica clinica, ricerca e sviluppo farmaceutico. I controlli di qualità molecolari servono a rilevare e correggere discrepanze, convalidare le prestazioni del test, aderire ai mandati normativi e, in ultima analisi, garantire la diffusione di risultati di test precisi e replicabili, aumentando così gli standard di assistenza ai pazienti e promuovendo la fiducia nei protocolli di test diagnostici.
La genomica è emersa come un aspetto fondamentale nell'avvento della medicina personalizzata , offrendo strategie terapeutiche e preventive su misura, adattate alle condizioni individuali per risultati ottimizzati. Questo paradigma medico è molto promettente nell'affrontare disturbi che finora hanno mostrato una risposta limitata ai trattamenti o ai rimedi convenzionali. Secondo il rapporto sulla medicina personalizzata pubblicato dalla Personalized Medicine Coalition, nel 2022, il Center for Drug Evaluation and Research (CDER) della FDA ha sanzionato 37 nuove entità molecolari (NME), con due NME designate come prodotti terapeutici e le restanti classificate come agenti diagnostici. Delle 35 nuove entità molecolari terapeutiche approvate dalla FDA statunitense, 12 erano interventi di medicina personalizzata, insieme a cinque nuove terapie basate sulle cellule.
| ATTRIBUTO | DETTAGLI |
| Periodo di studio | 2020-2029 |
| Anno base | 2021 |
| Anno stimato | 2022 |
| Anno previsto | 2022-2029 |
| Periodo storico | 2018-2020 |
| Unità | Valore (milioni di USD) |
| Segmentazione | Per prodotto, per applicazione, per tipo di analita, per settore di utilizzo finale, per regione |
| Per Prodotto |
|
| Per applicazione |
|
| Per tipo di analita |
|
| Per uso finale Industria |
|
| Per regione |
|
Negli ultimi sei anni, il costo del sequenziamento del genoma ha assistito a un rapido declino, stimato in una riduzione di circa un milione di volte. Il Progetto Genoma Umano, uno sforzo collaborativo che ha coinvolto migliaia di ricercatori nell'arco di 13 anni, ha sostenuto una spesa totale di 2,7 miliardi di USD, come documentato da WIRED nel settembre 2022. Un post del blog pubblicato su Sequencing in 2022 evidenzia il crescente interesse per il sequenziamento del DNA, spingendo numerose aziende bioinformatiche a concentrarsi su nuove tecnologie pionieristiche, piattaforme di sequenziamento e materiali di consumo con l'obiettivo di democratizzare i test genetici offrendoli a prezzi più accessibili.
[caption id="allegato_9614" align="aligncenter" width="870"]
Frequently Asked Questions
• What is the worth of global Molecular quality controls market?
The Molecular quality controls market size had crossed USD 154.43 Billion in 2020 and is expected to grow till USD 278.21 billion by 2029.
• Which is the largest regional market for Molecular quality controls market?
The Largest Regional market for Molecular quality controls market is APAC having a market share of 39%.
• What is the CAGR of Molecular quality controls market?
The global Molecular quality controls market registered a CAGR of 6.90% from 2022 to 2029.
• Which are the top companies to hold the market share in Molecular quality controls market?
Key players profiled in the report include F. Hoffmann-La Roche Ltd., Bio-Rad Laboratories, Inc., Thermo Fisher Scientific, Inc., Randox Laboratories Ltd., LGC Limited, Abbott Laboratories, Fortress Diagnostics, SERO AS, Anchor Molecular, Ortho-Clinical Diagnostics, Inc. , Quidel Corporation , Sun Diagnostics, LLC , Seegene Inc. , ZeptoMetrix, LLC, Qnostics ,Bio-Techne Corporation, Microbiologics, Inc., Microbix Biosystems Inc., SpeeDx Pty. Ltd., Maine Molecular Quality Controls, Inc., and Grifols, S.A.
Molecular Quality Controls Market Segment Analysis
The Molecular Quality Controls market is segmented by Product and service, Application, technology and end user.
On the basis of Product, the global Molecular Quality Control market is segmented into Independent, Instrument Specific (PCR, DNA Sequencing). The independent control segment held the greatest proportion of the global molecular quality control market. The expansion of the molecular quality control products market is being driven by an increase in the number of certified laboratories and regulatory mandates for the adoption of quality controls to ensure the accuracy of diagnostic test findings.
On the basis of Application, the global Molecular Quality Control market is segmented into Infectious Diseases Diagnostic, Oncology testing, genetic testing, others. The infectious disease diagnostics sector held the greatest proportion of the molecular quality controls market. The development of advanced assays for infectious diseases, a significant increase in the prevalence of infectious diseases, and rising awareness about the effective use of molecular diagnostic technologies to control the occurrence and spread of infectious diseases are all factors contributing to this segment's size.
On the basis of Analyte type, the global Molecular Quality Control market is segmented into Single-analyte Controls, Multi-analyte Controls. Due to the widespread use of single plex assays in hospitals and the benefits associated with single-analyte controls, such as straightforward analysis and interpretation and little risk of cross-reactivity, single-analyte controls dominated the molecular quality controls market in 2020.
On the basis of End use industry, the global Molecular Quality Control market is segmented into Diagnostic Laboratories, Hospitals, IVD Manufacturers & CROs, Academic and Research Institutes, Others.
[caption id="attachment_9572" align="alignleft" width="680"]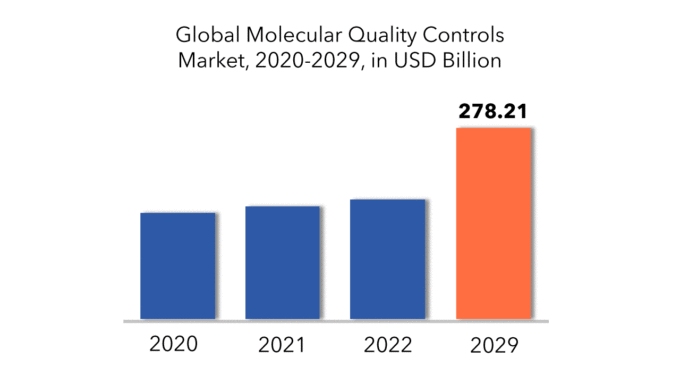
Molecular Quality Controls Market Players
The Key Players in Molecular Quality Controls market are F. Hoffmann-La Roche Ltd., Bio-Rad Laboratories, Inc., Thermo Fisher Scientific, Inc., Randox Laboratories Ltd. , LGC Limited , Abbott Laboratories , Fortress Diagnostics , SERO AS , Anchor Molecular, Ortho-Clinical Diagnostics, Inc. , Quidel Corporation , Sun Diagnostics, LLC , Seegene Inc. , ZeptoMetrix, LLC, Qnostics , Bio-Techne Corporation , Microbiologics, Inc., Microbix Biosystems Inc., SpeeDx Pty. Ltd., Maine Molecular Quality Controls, Inc., and Grifols, S.A..
In February 2020, The EDX RP Positive Run Control from Bio-Rad Laboratories (US) is the first multi-analyte control to target 22 analytes in a single vial, saving laboratories time and space on their equipment. AcroMetrix Coronavirus 2019 (COVID-19) RNA Control was also introduced by Thermo Fisher Scientific (US).
Who Should Buy? Or Key Stakeholders
- Industry and consulting firms
- State and national regulatory authorities
- Investors
- Suppliers and Distributors
- Research Institutes
- Others
Key Takeaways
- The global Molecular Quality Controls Market is expected to grow at a Compounded annual growth rate (CAGR) of 6.90%.
- On the basis of Product, the independent control segment held the greatest proportion of the global molecular quality control market.
- On the basis of Application, the infectious disease diagnostics sector held the greatest proportion of the molecular quality controls market.
- On the basis of Analyte type, single-analyte controls dominated the molecular quality controls market.
- On the basis of End use industry, Diagnostic Laboratories, Hospitals dominated the molecular quality controls market.
- North America was the largest regional market.
- Integration of advanced technologies such as CRISPR-based methods for precise molecular quality control assessments.
Molecular Quality Controls Market Regional Analysis
Geographically, Molecular Quality Controls market is segmented into North America, Europe, Asia Pacific, and Rest of the world.
- North America: includes the US, Canada, Mexico
- Europe: includes UK, Germany, France, Italy, Spain, Russia, and Rest of Europe
- Asia Pacific: includes China, Japan, South Korea, India, Australia, ASEAN and Rest of APAC
North America was the largest regional market for molecular quality controls due to the sophisticated healthcare systems in the United States and Canada, the existence of several major molecular quality control product manufacturers in the region, and easy access to technologically advanced goods.
The market in Asia Pacific, on the other hand, is expected to develop at the fastest rate over the projected period. The presence of rising economies like as China and India, as well as the strong need for high-quality and accurate diagnostic tests from the region's vast patient population, are driving the Asia Pacific molecular quality controls market forward.
[caption id="attachment_9615" align="alignnone" width="680"]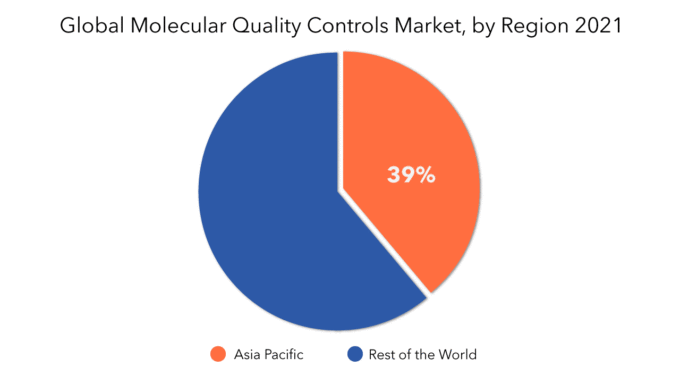
Key Market Segments: Molecular Quality Controls Market
Molecular Quality Controls Market by Product, 2020-2029 (USD Millions)- Independent
- Instrument Specific (PCR, DNA Sequencing)
- Infectious Diseases Diagnostic
- Oncology Testing
- Genetic Testing
- Others
- Single-Analyte Controls
- Multi-Analyte Controls
- Diagnostic Laboratories
- Hospitals
- IVD Manufacturers & CROS
- Academic And Research Institutes
- Others
- North America
- Europe
- APAC
- Mea
- Latin America
Objectives of the Study
- To provide an overall view of the market.
- To detail out key pointers such as growth, market size, trends, drivers, restraints, challenges and opportunities in quantitative and qualitative terms
- To provide a holistic view of market players and their activities to get an understanding of key growth strategies adopted
Key Question Answered
- What is the current market size of this high growth market?
- What is the overall growth rate?
- What are the key growth areas, applications, end uses and types?
- Key reasons for growth
- Challenges for growth
- What technological developments are happening in this area?
- Introduzione
- Definizione di mercato
- Segmentazione del mercato
- Cronologie della ricerca
- Presupposti e limitazioni
- Metodologia di ricerca
- Estrazione dei dati
- Ricerca secondaria
- Ricerca primaria
- Consigli degli esperti in materia
- Controlli di qualità
- Revisione finale
- Triangolazione dei dati
- Approccio dal basso verso l'alto
- Approccio dall'alto verso il basso
- Flusso di ricerca
- Fonti dei dati
- Estrazione dei dati
- Sintesi
- Panoramica del mercato
- Prospettive di mercato globali dei controlli di qualità molecolari
- Fattori trainanti del mercato
- Limitazioni di mercato
- Opportunità di mercato
- Impatto del Covid-19 sul mercato dei controlli di qualità molecolari
- Modello delle cinque forze di Porter
- Minaccia da parte dei nuovi entranti
- Minaccia da sostituti
- Potere contrattuale dei fornitori
- Potere contrattuale dei clienti
- Grado di concorrenza
- Analisi della catena del valore del settore
- Prospettive di mercato globali dei controlli di qualità molecolari
- Mercato globale dei controlli di qualità molecolari per prodotto
- Indipendente
- Strumento specifico (PCR, sequenziamento del DNA)
- Mercato globale dei controlli di qualità molecolari per applicazione
- Diagnosi delle malattie infettive
- Test oncologici
- Test genetici
- Altri
- Mercato globale dei controlli di qualità molecolari per tipo di analita
- Controlli a singolo analita
- controlli multi-analita
- Mercato globale dei controlli di qualità molecolari per settore di utilizzo finale
- Laboratori diagnostici
- Ospedali
- Produttori IVD e CROS
- Istituti accademici e di ricerca
- Altri
- Mercato globale dei controlli di qualità molecolari per regione
- America del Nord
- NOI
- Canada
- Messico
- Sud America
- Brasile
- Argentina
- Colombia
- Resto del Sud America
- Europa
- Germania
- Regno Unito
- Francia
- Italia
- Spagna
- Russia
- Resto d'Europa
- Asia Pacifico
- India
- Cina
- Giappone
- Corea del Sud
- Australia
- Asia sud-orientale
- Resto dell'Asia Pacifica
- Medio Oriente e Africa
- Emirati Arabi Uniti
- Arabia Saudita
- Sudafrica
- Resto del Medio Oriente e Africa
- America del Nord
- Profili aziendali* (panoramica aziendale, istantanea aziendale, tipi di analiti offerti, sviluppi recenti)
- Hoffmann-La Roche Ltd.,
- Laboratori Bio-Rad, Inc.,
- Thermo Fisher Scientific, Inc.,
- Laboratori Randox Ltd,
- LGC limitata
- Laboratori Abbott
- Diagnostica della Fortezza
- SIERO COME
- Ancora molecolare
- Diagnostica orto-clinica, Inc.
- Società Quidel
- Diagnostica solare LLC
- Seegene Inc. *L'elenco delle aziende è indicativo
ELENCO DELLE TABELLE
TABLE 1 GLOBAL MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 2 GLOBAL MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 3 GLOBAL MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 4 GLOBAL MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 5 GLOBAL MOLECULAR QUALITY CONTROLS MARKET BY REGION (USD MILLIONS) 2020-2029
TABLE 6 US MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 7 US MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 8 US MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 9 US MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 10 CANADA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 11 CANADA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 12 CANADA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 13 CANADA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 14 MEXICO MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 15 MEXICO MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 16 MEXICO MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 17 MEXICO MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 18 BRAZIL MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 19 BRAZIL MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 20 BRAZIL MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 21 BRAZIL MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 22 ARGENTINA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 23 ARGENTINA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 24 ARGENTINA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 25 ARGENTINA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 26 COLOMBIA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 27 COLOMBIA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 28 COLOMBIA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 29 COLOMBIA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 30 REST OF SOUTH AMERICA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 31 REST OF SOUTH AMERICA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 32 REST OF SOUTH AMERICA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 33 REST OF SOUTH AMERICA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 34 INDIA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 35 INDIA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 36 INDIA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 37 INDIA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 38 CHINA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 39 CHINA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 40 CHINA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 41 CHINA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 42 JAPAN MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 43 JAPAN MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 44 JAPAN MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 45 JAPAN MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 46 SOUTH KOREA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 47 SOUTH KOREA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 48 SOUTH KOREA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 49 SOUTH KOREA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 50 AUSTRALIA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 51 AUSTRALIA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 52 AUSTRALIA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 53 AUSTRALIA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 54 SOUTH-EAST ASIA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 55 SOUTH-EAST ASIA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 56 SOUTH-EAST ASIA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 57 SOUTH-EAST ASIA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 58 REST OF ASIA PACIFIC MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 59 REST OF ASIA PACIFIC MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 60 REST OF ASIA PACIFIC MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 61 REST OF ASIA PACIFIC MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 62 GERMANY MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 63 GERMANY MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 64 GERMANY MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 65 GERMANY MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 66 UK MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 67 UK MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 68 UK MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 69 UK MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 70 FRANCE MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 71 FRANCE MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 72 FRANCE MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 73 FRANCE MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 74 ITALY MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 75 ITALY MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 76 ITALY MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 77 ITALY MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 78 SPAIN MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 79 SPAIN MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 80 SPAIN MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 81 SPAIN MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 82 RUSSIA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 83 RUSSIA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 84 RUSSIA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 85 RUSSIA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 86 REST OF EUROPE MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 87 REST OF EUROPE MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 88 REST OF EUROPE MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 89 REST OF EUROPE MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 90 UAE MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 91 UAE MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 92 UAE MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 93 UAE MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 94 SAUDI ARABIA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 95 SAUDI ARABIA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 96 SAUDI ARABIA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 97 SAUDI ARABIA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 98 SOUTH AFRICA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 99 SOUTH AFRICA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 100 SOUTH AFRICA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 101 SOUTH AFRICA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
TABLE 102 REST OF MIDDLE EAST AND AFRICA MOLECULAR QUALITY CONTROLS MARKET BY PRODUCT (USD MILLIONS) 2020-2029
TABLE 103 REST OF MIDDLE EAST AND AFRICA MOLECULAR QUALITY CONTROLS MARKET BY APPLICATION (USD MILLIONS) 2020-2029
TABLE 104 REST OF MIDDLE EAST AND AFRICA MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE (USD MILLIONS) 2020-2029
TABLE 105 REST OF MIDDLE EAST AND AFRICA MOLECULAR QUALITY CONTROLS MARKET BY END USER (USD MILLIONS) 2020-2029
LIST OF FIGURES
FIGURE 1 MARKET DYNAMICS
FIGURE 2 MARKET SEGMENTATION
FIGURE 3 REPORT TIMELINES: YEARS CONSIDERED
FIGURE 4 DATA TRIANGULATION
FIGURE 5 BOTTOM-UP APPROACH
FIGURE 6 TOP-DOWN APPROACH
FIGURE 7 RESEARCH FLOW
FIGURE 8 GLOBAL MOLECULAR QUALITY CONTROLS MARKET BY ANALYTE TYPE & SERVICE, USD MILLION, 2020-2029
FIGURE 9 GLOBAL MOLECULAR QUALITY CONTROLS MARKET BY MATERIAL, USD MILLION, 2020-2029
FIGURE 10 GLOBAL MOLECULAR QUALITY CONTROLS MARKET BY MODULES, USD MILLION, 2020-2029
FIGURE 11 GLOBAL MOLECULAR QUALITY CONTROLS MARKET BY END USE INDUSTRY, USD MILLION, 2020-2029
FIGURE 12 GLOBAL MOLECULAR QUALITY CONTROLS MARKET BY REGION, USD MILLION, 2020-2029
FIGURE 13 PORTER’S FIVE FORCES MODEL
FIGURE 14 NORTH AMERICA MOLECULAR QUALITY CONTROLS MARKET SNAPSHOT
FIGURE 15 EUROPE MOLECULAR QUALITY CONTROLS MARKET SNAPSHOT
FIGURE 16 ASIA PACIFIC MOLECULAR QUALITY CONTROLS MARKET SNAPSHOT
FIGURE 17 SOUTH AMERICA MOLECULAR QUALITY CONTROLS MARKET SNAPSHOT
FIGURE 18 MIDDLE EAST & AFRICA MOLECULAR QUALITY CONTROLS MARKET SNAPSHOT
FIGURE 19 F. Hoffmann-La Roche Ltd., COMPANY SNAPSHOT
FIGURE 20 BIO-RAD LABORATORIES, INC., COMPANY SNAPSHOT
FIGURE 21 THERMO FISHER SCIENTIFIC, INC., COMPANY SNAPSHOT
FIGURE 22 RANDOX LABORATORIES LTD, COMPANY SNAPSHOT
FIGURE 23 LGC LIMITED COMPANY SNAPSHOT
FIGURE 24 ABBOTT LABORATORIES COMPANY SNAPSHOT
FIGURE 25 FORTRESS DIAGNOSTICS COMPANY SNAPSHOT
FIGURE 26 SERO AS COMPANY SNAPSHOT
FIGURE 27 ANCHOR MOLECULAR COMPANY SNAPSHOT
FIGURE 28 ORTHO-CLINICAL DIAGNOSTICS, INC. COMPANY SNAPSHOT
FIGURE 29 QUIDEL CORPORATION COMPANY SNAPSHOT
FIGURE 30 SUN DIAGNOSTICS LLC COMPANY SNAPSHOT
FIGURE 31 SEEGENE INC. COMPANY SNAPSHOT
DOWNLOAD FREE SAMPLE REPORT
License Type
SPEAK WITH OUR ANALYST
Want to know more about the report or any specific requirement?
WANT TO CUSTOMIZE THE REPORT?
Our Clients Speak
We asked them to research ‘ Equipment market’ all over the world, and their whole arrangement was helpful to us. thehealthanalytics.com insightful analysis and reports contributed to our current analysis and in creating a future strategy. Besides, the te
Yosuke Mitsui
Senior Associate Construction Equipment Sales & Marketing
We asked them to research ‘Equipment market’ all over the world, and their whole arrangement was helpful to us. thehealthanalytics.com insightful analysis and reports contributed to our current analysis and in creating a future strategy. Besides, the te






