
Base Year Value ()
CAGR ()
Forecast Year Value ()
Historical Data Period
Largest Region
Forecast Period
Mercato degli archivi indipendenti dal fornitore per tipo (PACS, software VNA), modalità di distribuzione (in loco, ibrida, ospitata nel cloud), modello di utilizzo (singoli reparti, più reparti, più siti) e per regione (Nord America, Europa, Asia Pacifico, Sud America, Medio Oriente e Africa), tendenze globali e previsioni dal 2022 al 2029
Instant access to hundreds of data points and trends
- Market estimates from 2014-2029
- Competitive analysis, industry segmentation, financial benchmarks
- Incorporates SWOT, Porter's Five Forces and risk management frameworks
- PDF report or online database with Word, Excel and PowerPoint export options
- 100% money back guarantee
Panoramica del mercato degli archivi neutrali rispetto al fornitore
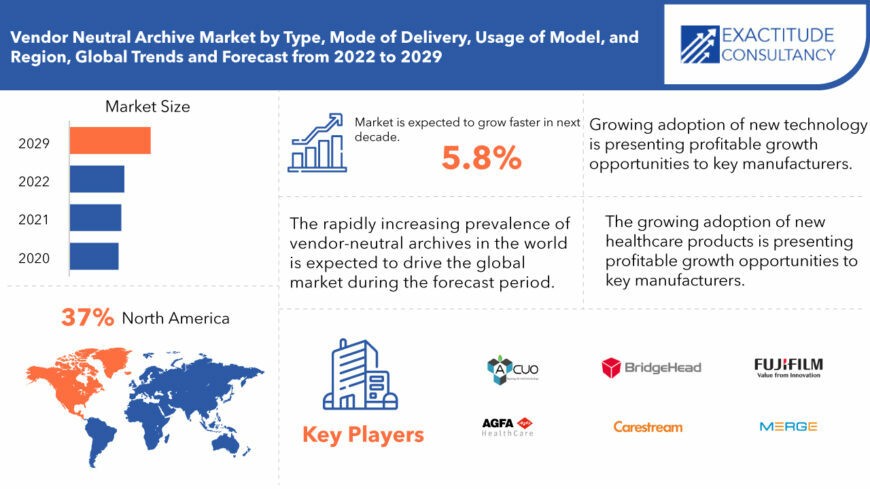
Si prevede che il mercato globale degli archivi vendor-neutral crescerà a un CAGR superiore al 5,8% dal 2022 al 2029. Si prevede che raggiungerà oltre 622 milioni di USD entro il 2029 dai 374 milioni di USD del 2020.
L'espansione osservata in questo mercato può essere attribuita a diversi fattori, tra cui il numero sostanziale di approvazioni concesse per i dispositivi di chiusura vascolare, l'incidenza prevalente di malattie cardiovascolari, la crescente prevalenza di obesità, la crescente enfasi delle entità di mercato sui dispositivi di chiusura vascolare di grande calibro, la crescente demografia geriatrica, il favorevole panorama di rimborso per le procedure di radiologia interventistica e l'attenzione accresciuta sulla gestione efficace della perdita di sangue durante gli interventi chirurgici. Inoltre, i mercati emergenti presentano prospettive di crescita redditizie per i partecipanti al mercato, insieme alla proliferazione di ospedali e centri chirurgici, che collettivamente offrono solide opportunità di crescita. Tuttavia, sfide come il requisito di professionisti altamente qualificati, i rischi intrinseci associati ai dispositivi di chiusura vascolare e il rigido quadro normativo possono ostacolare in una certa misura l'espansione del mercato. Il mercato dei dispositivi di chiusura vascolare è segmentato in base a tipo, accesso, procedura e regione.
Il mercato dei dispositivi di chiusura vascolare (VCD) comprende una serie di dispositivi medici progettati per ottenere l'emostasi in seguito a procedure vascolari minimamente invasive, in particolare cateterizzazione cardiaca e interventi vascolari periferici. Questi dispositivi sono fondamentali per sigillare i siti di puntura all'interno di arterie o vene, mitigando così il rischio di complicazioni emorragiche post-procedurali. I VCD presentano vantaggi distinti rispetto alle tecniche di compressione manuale convenzionali, tra cui emostasi accelerata, tempi di deambulazione ridotti, minore disagio per il paziente e ricoveri ospedalieri abbreviati.
| ATTRIBUTO | DETTAGLI |
| Periodo di studio | 2020-2029 |
| Anno base | 2021 |
| Anno stimato | 2022 |
| Anno previsto | 2022-2029 |
| Periodo storico | 2018-2020 |
| Unità | Valore (miliardi di USD) |
| Segmentazione | Per tipo, per modalità di consegna, utilizzo del modello |
| Per tipo |
|
| Per modalità di consegna |
|
| Per utilizzo del modello |
|
| Per regione |
|
Il panorama normativo globale che disciplina i dispositivi di chiusura vascolare è caratterizzato da un quadro multiforme che comprende standard, linee guida e regolamenti promulgati dalle autorità di regolamentazione in diverse nazioni e regioni. Enti come la Food and Drug Administration (FDA) degli Stati Uniti, l'Agenzia europea per i medicinali (EMA) e la Japan Pharmaceuticals and Medical Devices Agency (PMDA) delineano i requisiti relativi alla sicurezza, all'efficacia e alla qualità dei VCD tramite processi di approvazione pre-commercializzazione, meccanismi di sorveglianza post-commercializzazione e valutazioni di routine.
[caption id="allegato_6568" align="aligncenter" width="870"]
Frequently Asked Questions
• What is the worth of the global vendor neutral archive market?
The vendor neutral archive market size has crossed USD 374 million in 2020 and will observe a CAGR of more than 5.8% up to 2027 driven by the rising technological advancements in molecular biology, genomics, and microbial diversity.
• What is the size of the North America vendor neutral archive industry?
North America held more than 37% of the vendor neutral archive market revenue share in 2020 and will witness expansion with the surging demand for vendor neutral archive in countries, including U.S. and Canada.
• What are the upcoming trends of the vendor neutral archive market, globally?
The upcoming trends in the vendor neutral archive market are expansion in the technological advancements in vendor neutral archive will drive industry revenue in the next few years
• What is the CAGR of the vendor neutral archive market?
The global vendor neutral archive market registered a CAGR of 5.8% from 2022 to 2029. The application segment was the highest revenue contributor to the market, with 374 million in 2020, and is estimated to reach 622 million by 2029, with a CAGR of 7%.
• Which are the top companies to hold the market share in the vendor neutral archive market?
Key players profiled in the report include Acuo Technologies (U.S.), Agfa HealthCare (Belgium), BridgeHead Software (U.K.), Carestream Health (U.S.), Dell, Inc. (U.S.), GE Healthcare (U.K.), Fujifilm Corporation (Japan), Merge Health (U.S.), McKesson Corporation (U.S.), Philips Healthcare (The Netherlands), Siemens Healthcare (Germany), and TeraMedica (U.S.).
• What is the leading application of the vendor neutral archive market?
The healthcare application is expected to increase the most.
• Which is the largest regional market for vendor neutral archive market?
North America is the largest regional market for vendor neutral archive.
Vendor Neutral Archive Market Segment Analysis
The two key segments of the PACS market are departmental PACS and enterprise PACS. The vendor-neutral archive market is divided into three types: passive approximators, active approximators, and external hemostatic devices. The market for the vendor-neutral archive is dominated by passive approximators. The growing number of vascular closure device approvals, as well as the numerous benefits connected with these devices, such as ease of use and a lower complication rate in patients, can be attributable to the substantial proportion of this market. Along with the worldwide VNA market, the PACS market is included to provide a comparative overview of both healthcare information systems. The product lifecycles of each of these systems differ greatly. The vendor-neutral archives (VNA) market is thought to be in the same phase (exponential phase) as the PACS market was 10 to 15 years ago. The PACS market, on the other hand, is a mature industry defined by market participants attempting to invest in product innovation and seeking new consumer groups.
The market is divided into three segments based on delivery mode: on-premise VNAs, hybrid VNAs, and entirely cloud-hosted VNAs. Because this approach is preferred over others, on-premise VNAs currently dominate the industry in terms of overall revenue contribution. This choice is due to the superior data control and security afforded by on-premise VNA solutions. However, due to the high capital investment required for on-premise VNAs, hybrid VNA systems that include a cloud-based component for storage are currently attracting the interest of small and medium-sized healthcare facilities. As a result, hybrid VNA solutions are expected to grow faster than on-premise solutions.
The benefits of vendor-neutral archives over PACS in terms of interoperability, flexibility, and accessibility are driving market adoption of VNA technology. In addition, favorable and promising government reforms in the sector of healthcare information technology are encouraging the market. Rising data breaches, the complexity of the VNA idea, the lack of a proper industry-accepted definition of VNAs, and long VNA lifecycles are the key impediments to market expansion (that impact VNA deployment decisions). As a result, the market is poised to witness trends such as VNA integration with EHRs, the use of cloud computing solutions for VNA products, and a holistic approach to providing superior patient care.Picture archiving and communication systems (PACS) are one of the most extensively used technologies in hospitals around the world. PACS is used in more than 80% of hospitals in affluent countries such as the United States, the United Kingdom, and Japan. The scope is visible in developing countries, notably in Asian countries such as India and China. These countries' healthcare facilities are now transitioning from the usage of different services to integrated systems for sharing and storing medical pictures. PACS adoption is fast expanding in these countries as healthcare sectors improve, healthcare expenditure rises, and people become more aware of the need for picture archiving and communication systems. These technologies are generally used in radiology.
The benefit of an active vendor-neutral archive in shortening time to ambulation and hemostasis commanded more than 58 percent of revenue share in 2020. The active vascular closure device employs a variety of ways for direct closure of the artery entry site, including the use of collagen or suture-based materials. Increasing product availability to a bigger population opens up revenue generation opportunities for the segment. Collagen plug devices such as Angio-seal and Mynx, as well as ExoSeal and Starclose devices, are examples of these devices. As a result, more product releases by market participants will benefit segment growth.
Picture archiving and communication systems (PACS) are one of the most extensively used technologies in hospitals around the world. PACS is used in more than 80% of hospitals in affluent countries such as the United States, the United Kingdom, and Japan. The scope is visible in developing countries, notably in Asian countries such as India and China. These countries' healthcare facilities are now transitioning from the usage of different services to integrated systems for sharing and storing medical pictures. PACS adoption is fast expanding in these countries as healthcare sectors improve, healthcare expenditure rises, and people become more aware of the need for picture archiving and communication systems. These technologies are generally used in radiology.
[caption id="attachment_6572" align="aligncenter" width="680"]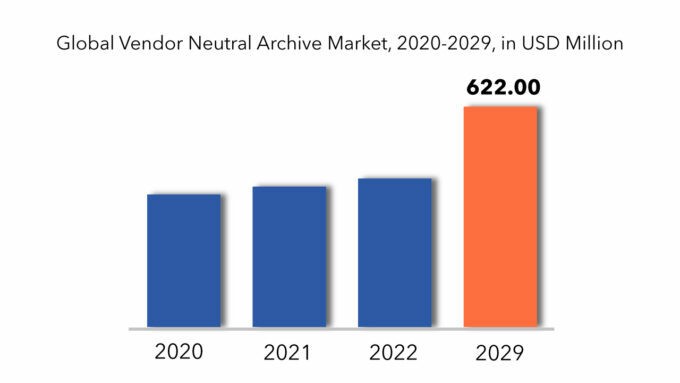
Vendor Neutral Archive Market Key Players
The prominent players in the vendor-neutral archive market include Acuo Technologies (U.S.), Agfa HealthCare (Belgium), BridgeHead Software (U.K.), Carestream Health (U.S.), Dell, Inc. (U.S.), GE Healthcare (U.K.), Fujifilm Corporation (Japan), Merge Health (U.S.), McKesson Corporation (U.S.), Philips Healthcare (The Netherlands), Siemens Healthcare (Germany), and TeraMedica (U.S.).
Who Should Buy? Or Key stakeholders
- Investors
- Environment, Health, and Safety Professionals
- Research Organizations
- Medical industry
- Regulatory Authorities
- Others
Key Takeaways
- The global vendor-neutral archive market is expected to grow at more than 5.8% CAGR.
- The market for the vendor-neutral archive is dominated by passive approximators.
- Based on delivery mode, on-premise VNAs currently dominate the industry.
- The North America market is predicted to lead the global vendor neutral archive market.
- The major trend in the Vendor Neutral Archive (VNA) market is the integration of artificial intelligence (AI) for advanced data analytics and workflow optimization.
Vendor Neutral Archive Market Regional Analysis
The vendor-neutral archive market is divided into major regions- North America, Europe, South America, Asia Pacific, and middle east & Africa. North America is the largest regional segment in 2021.
- North America: includes the US, Canada, Mexico
- Europe: includes UK, Germany, France, Italy, Spain, Russia, and Rest of Europe
- Asia Pacific: includes China, Japan, South Korea, India, Australia, ASEAN and Rest of APAC
- South America: includes Brazil, Argentina and Rest of South America
- Middle East & Africa: includes Turkey, UAE, Saudi Arabia, South Africa, and Rest of MEA
When compared to other regional markets, the North America market is predicted to lead the global vendor neutral archive market with a greater revenue share, and this dominance is expected to continue during the forecast period. This might be ascribed to the region's well-developed healthcare industry and the presence of important market participants. Europe has the second-largest revenue share in the global vendor neutral archive market, followed by Asia Pacific, Latin America, and the Middle East and Africa, in that order. The Asia Pacific market is expected to grow the fastest in terms of revenue, owing to increased adoption of imaging in various departments such as cardiology, pathology, oncology, and dermatology, as well as increased government initiatives for the development of healthcare sector in emerging markets.
[caption id="attachment_6573" align="aligncenter" width="680"]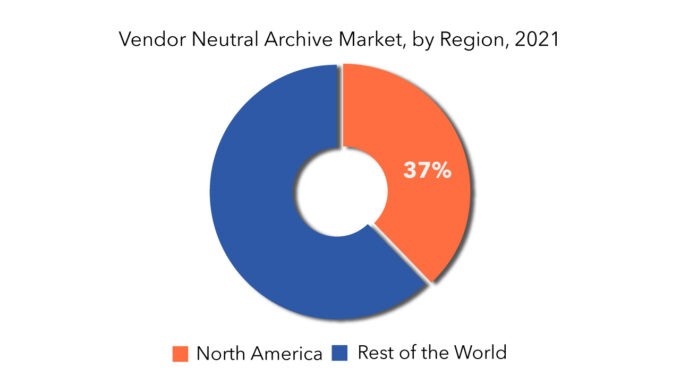
Key Market Segments: Global vascular closure devices Market
Vendor Neutral Archive Market by Type, 2020-2029, (USD Million)- PACS
- VNA Software
- On-Site
- Hybrid
- Cloud-Hosted
- Single Department
- Multiple Department
- Multiple Sites
- North America
- Europe
- Asia Pacific
- South America
- Middle East And Africa
Key Question Answered
- What is the current market size of this high growth market?
- What is the overall growth rate?
- What are the key growth areas, types, Applications?
- Key reasons for growth
- Challenges for growth
- What technological developments are happening in this area?
- INTRODUZIONE
- DEFINIZIONE DI MERCATO
- SEGMENTAZIONE DEL MERCATO
- TEMPI DI RICERCA
- ASSUNZIONI E LIMITAZIONI
- METODOLOGIA DI RICERCA
- ESTRAZIONE DATI
- RICERCA SECONDARIA
- RICERCA PRIMARIA
- PARERI DEGLI ESPERTI IN MATERIA
- CONTROLLI DI QUALITÀ
- RECENSIONE FINALE
- TRIANGOLAZIONE DEI DATI
- APPROCCIO DAL BASSO VERSO L'ALTO
- APPROCCIO TOP-DOWN
- FLUSSO DI RICERCA
- FONTI DEI DATI
- ESTRAZIONE DATI
- SINTESI
- PANORAMICA DEL MERCATO
- PROSPETTIVE DI MERCATO ARCHIVIAZIONE NEUTRALE PER I FORNITORI GLOBALI
- FATTORI DI MERCATO
- LIMITAZIONI DI MERCATO
- OPPORTUNITÀ DI MERCATO
- IMPATTO DEL COVID-19 SUL MERCATO DEGLI ARCHIVI NEUTRI PER I FORNITORI
- MODELLO DELLE CINQUE FORZE DI PORTER
- MINACCIA DAI NUOVI ENTRANSI
- MINACCIA DA SOSTITUTI
- POTERE CONTRATTUALE DEI FORNITORI
- POTERE CONTRATTUALE DEI CLIENTI
- GRADO DI COMPETIZIONE
- ANALISI DELLA CATENA DEL VALORE DEL SETTORE
- PROSPETTIVE DI MERCATO ARCHIVIAZIONE NEUTRALE PER I FORNITORI GLOBALI
- MERCATO ARCHIVIAZIONE GLOBALE NEUTRALE PER TIPOLOGIA, (MILIONI DI USD)
- PACCHI
- SOFTWARE VNA
- MERCATO ARCHIVIAZIONE GLOBALE NEUTRALE PER FORNITORE PER MODALITÀ DI CONSEGNA, (MILIONI DI USD)
- SUL POSTO
- IBRIDO
- OSPITATO NEL CLOUD
- MERCATO ARCHIVIAZIONE GLOBALE NEUTRALE PER FORNITORE PER UTILIZZO DEL MODELLO (MILIONI DI USD)
- DIPARTIMENTO SINGOLO
- DIPARTIMENTO MULTIPLO
- SITI MULTIPLI
- MERCATO ARCHIVIO NEUTRO PER FORNITORI GLOBALI PER REGIONE
- AMERICA DEL NORD
- NOI
- CANADA
- MESSICO
- SUD AMERICA
- BRASILE
- ARGENTINA
- COLOMBIA
- RESTO DEL SUD AMERICA
- EUROPA
- GERMANIA
- Regno Unito
- FRANCIA
- ITALIA
- SPAGNA
- RUSSIA
- RESTO D'EUROPA
- ASIA PACIFICO
- INDIA
- CINA
- GIAPPONE
- COREA DEL SUD
- AUSTRALIA
- ASIA SUD-ORIENTALE
- RESTO DELL'ASIA PACIFICO
- MEDIO ORIENTE E AFRICA
- Emirati Arabi Uniti
- ARABIA SAUDITA
- SUDAFRICA
- RESTO DEL MEDIO ORIENTE E AFRICA
- AMERICA DEL NORD
- PROFILI AZIENDALI* (PANORAMICA AZIENDALE, PANORAMICA AZIENDALE, PRODOTTI OFFERTI, SVILUPPI RECENTI)
- TECNOLOGIE ACUO
- AGFA SANITÀ
- SOFTWARE TESTA DI PONTE
- SALUTE CARESTREAM
- DELL, INC.
- ASSISTENZA SANITARIA GE
- Società Fujifilm
- UNISCI SALUTE
- CORPORAZIONE MCKESSON
- ASSISTENZA SANITARIA PHILIPS
- ASSISTENZA SANITARIA SIEMENS
- TERAMEDICA (USA). CHIRURGIA INTUITIVA
- ALTRI GIOCATORI *L'ELENCO DELLE SOCIETÀ È INDICATIVO
ELENCO DELLE TABELLE
TABLE 1 GLOBAL VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 2 GLOBAL VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 3 GLOBAL VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 4 GLOBAL VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 5 GLOBAL VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 6 GLOBAL VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 7 GLOBAL VENDOR NEUTRAL ARCHIVE MARKET BY REGION (USD MILLIONS) 2020-2029
TABLE 8 GLOBAL VENDOR NEUTRAL ARCHIVE MARKET BY REGION 2020-2029
TABLE 9 US VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 10 US VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 11 US VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 12 US VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 13 US VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 14 US VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 15 CANADA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 16 CANADA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 17 CANADA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 18 CANADA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 19 CANADA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 20 CANADA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 21 MEXICO VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 22 MEXICO VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 23 MEXICO VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 24 MEXICO VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 25 MEXICO VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 26 MEXICO VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 27 BRAZIL VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 28 BRAZIL VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 29 BRAZIL VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 30 BRAZIL VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 31 BRAZIL VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 32 BRAZIL VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 33 ARGENTINA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 34 ARGENTINA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 35 ARGENTINA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 36 ARGENTINA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 37 ARGENTINA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 38 ARGENTINA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 39 COLOMBIA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 40 COLOMBIA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 41 COLOMBIA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 42 COLOMBIA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 43 COLOMBIA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 44 COLOMBIA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 45 REST OF SOUTH AMERICA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 46 REST OF SOUTH AMERICA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 47 REST OF SOUTH AMERICA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 48 REST OF SOUTH AMERICA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 49 REST OF SOUTH AMERICA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 50 REST OF SOUTH AMERICA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 51 INDIA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 52 INDIA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 53 INDIA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 54 INDIA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 55 INDIA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 56 INDIA VENDOR NEUTRAL ARCHIVE MARKET BY END-USER 2020-2029
TABLE 57 CHINA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 58 CHINA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 59 CHINA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 60 CHINA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 61 CHINA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 62 CHINA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 63 JAPAN VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 64 JAPAN VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 65 JAPAN VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 66 JAPAN VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 67 JAPAN VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 68 JAPAN VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 69 SOUTH KOREA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 70 SOUTH KOREA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 71 SOUTH KOREA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 72 SOUTH KOREA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 73 SOUTH KOREA M MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 74 SOUTH KOREA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 75 AUSTRALIA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 76 AUSTRALIA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 77 AUSTRALIA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 78 AUSTRALIA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 79 AUSTRALIA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 80 AUSTRALIA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 81 SOUTH-EAST ASIA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 82 SOUTH-EAST ASIA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 83 SOUTH-EAST ASIA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 84 SOUTH-EAST ASIA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 85 SOUTH-EAST ASIA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 86 SOUTH-EAST ASIA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 87 REST OF ASIA PACIFIC VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 88 REST OF ASIA PACIFIC VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 89 REST OF ASIA PACIFIC VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 90 REST OF ASIA PACIFIC VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 91 REST OF ASIA PACIFIC VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 92 REST OF ASIA PACIFIC VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 93 GERMANY VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 94 GERMANY VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 95 GERMANY VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 96 GERMANY VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 97 GERMANY VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 98 GERMANY VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 99 UK VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 100 UK VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 101 UK VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 102 UK VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 103 UK VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 104 UK VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 105 FRANCE VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 106 FRANCE VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 107 FRANCE VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 108 FRANCE VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 109 FRANCE VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 110 FRANCE VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 111 ITALY VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 112 ITALY VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 113 ITALY VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 114 ITALY VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 115 ITALY VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 116 ITALY VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 117 SPAIN VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 118 SPAIN VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 119 SPAIN VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 120 SPAIN VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 121 SPAIN VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 122 SPAIN VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 123 RUSSIA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 124 RUSSIA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 125 RUSSIA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 126 RUSSIA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 127 RUSSIA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 128 RUSSIA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 129 REST OF EUROPE VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 130 REST OF EUROPE VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 131 REST OF EUROPE VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 132 REST OF EUROPE VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 133 REST OF EUROPE VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 134 REST OF EUROPE VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 135 UAE VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 136 UAE VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 137 UAE VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 138 UAE VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 139 UAE VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 140 UAE VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 141 SAUDI ARABIA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 142 SAUDI ARABIA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 143 SAUDI ARABIA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 144 SAUDI ARABIA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 145 SAUDI ARABIA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 146 SAUDI ARABIA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 147 SOUTH AFRICA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 148 SOUTH AFRICA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 149 SOUTH AFRICA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 150 SOUTH AFRICA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 151 SOUTH AFRICA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 152 SOUTH AFRICA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
TABLE 153 REST OF MIDDLE EAST AND AFRICA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE(USD MILLIONS) 2020-2029
TABLE 154 REST OF MIDDLE EAST AND AFRICA VENDOR NEUTRAL ARCHIVE MARKET BY TYPE 2020-2029
TABLE 155 REST OF MIDDLE EAST AND AFRICA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY (USD MILLIONS) 2020-2029
TABLE 156 REST OF MIDDLE EAST AND AFRICA VENDOR NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY 2020-2029
TABLE 157 REST OF MIDDLE EAST AND AFRICA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL(USD MILLIONS) 2020-2029
TABLE 158 REST OF MIDDLE EAST AND AFRICA VENDOR NEUTRAL ARCHIVE MARKET BY USAGE OF MODEL 2020-2029
LIST OF FIGURES
FIGURE 1 MARKET DYNAMICS
FIGURE 2 MARKET SEGMENTATION
FIGURE 3 REPORT TIMELINES: YEARS CONSIDERED
FIGURE 4 DATA TRIANGULATION
FIGURE 5 BOTTOM-UP APPROACH
FIGURE 6 TOP-DOWN APPROACH
FIGURE 7 RESEARCH FLOW
FIGURE 8 GLOBAL VENDORS NEUTRAL ARCHIVE MARKET BY VEHICLE TYPE, USD MILLION, 2020-2029
FIGURE 9 GLOBAL VENDORS NEUTRAL ARCHIVE MARKET BY MODE OF DELIVERY, USD MILLION, 2020-2029
FIGURE 10 GLOBAL VENDORS NEUTRAL ARCHIVE MARKET BY END USER, USD MILLION, 2020-2029
FIGURE 11 GLOBAL VENDORS NEUTRAL ARCHIVE MARKET BY REGION, USD MILLION, 2020-2029
FIGURE 12 PORTER’S FIVE FORCES MODEL
FIGURE 13 NORTH AMERICA VENDOR NEUTRAL ARCHIVE MARKET SNAPSHOT
FIGURE 14 EUROPE VENDOR NEUTRAL ARCHIVE MARKET SNAPSHOT
FIGURE 15 ASIA PACIFIC VENDOR NEUTRAL ARCHIVE MARKET SNAPSHOT
FIGURE 16 SOUTH AMERICA VENDOR NEUTRAL ARCHIVE MARKET SNAPSHOT
FIGURE 17 MIDDLE EAST & AFRICA VENDOR NEUTRAL ARCHIVE MARKET SNAPSHOT
FIGURE 18 ACUO TECHNOLOGIES: COMPANY SNAPSHOT
FIGURE 19 AGFA HEALTHCARE: COMPANY SNAPSHOT
FIGURE 20 BRIDGEHEAD SOFTWARE : COMPANY SNAPSHOT
FIGURE 21 CARESTREAM HEALTH: COMPANY SNAPSHOT
FIGURE 22 DELL, INC. : COMPANY SNAPSHOT
FIGURE 23 GE HEALTHCARE : COMPANY SNAPSHOT
FIGURE 24 FUJIFILM CORPORATION : COMPANY SNAPSHOT
FIGURE 25 MERGE HEALTH: COMPANY SNAPSHOT
FIGURE 26 MCKESSON CORPORATION : COMPANY SNAPSHOT
FIGURE 27 PHILIPS HEALTHCARE: COMPANY SNAPSHOT
FIGURE 28 SIEMENS HEALTHCARE: COMPANY SNAPSHOT
FIGURE 29 TERAMEDICA: COMPANY SNAPSHOT
DOWNLOAD FREE SAMPLE REPORT
License Type
SPEAK WITH OUR ANALYST
Want to know more about the report or any specific requirement?
WANT TO CUSTOMIZE THE REPORT?
Our Clients Speak
We asked them to research ‘ Equipment market’ all over the world, and their whole arrangement was helpful to us. thehealthanalytics.com insightful analysis and reports contributed to our current analysis and in creating a future strategy. Besides, the te
Yosuke Mitsui
Senior Associate Construction Equipment Sales & Marketing
We asked them to research ‘Equipment market’ all over the world, and their whole arrangement was helpful to us. thehealthanalytics.com insightful analysis and reports contributed to our current analysis and in creating a future strategy. Besides, the te






